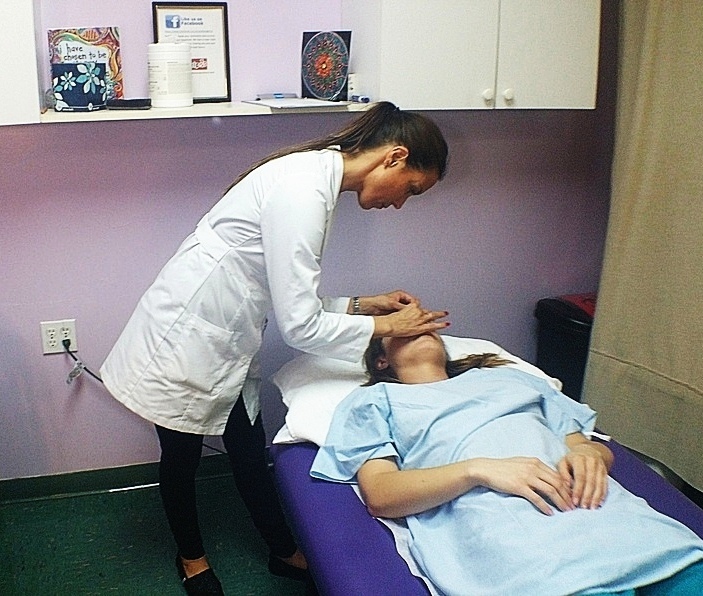
If you’re reading this, then you’ve likely had acupuncture treatments in the past, realized how great Traditional Chinese Medicine (TCM) is, and are now thinking about studying acupuncture. … Read More
Ultimate Guide for Choosing an Acupuncture School
We Asked OpenAI GPT-3: How do I succeed as a massage therapist?
By now, you've likely heard of OpenAI GPT-3, which is an artificial intelligence language platform that can produce human-like text. … Read More
Shiatsu Career Information | Acupuncture and Massage College | Miami, FL
Over the past few weeks, we have been discussing Shiatsu. Shiatsu is a form of Japanese massage that is based on the principles of Traditional Chinese Medicine (TCM), and it is considered to be a form of acupressure massage. However, unlike other forms of acupressure massage, Shiatsu exclusively utilizes finger and palm massage techniques to treat a patient’s pain and health conditions. … Read More
History of Shiatsu | Acupuncture and Massage College | Miami, FL
Shiatsu is a core modality taught at our massage school. In the classroom, we mainly focus on teaching the application of the techniques of shiatsu. However, we think that this modality is so fascinating that it deserves its own blog series. In an earlier post, we gave an overview of Shiatsu massage. In this post, we will discuss the history of Shiatsu massage. Shiatsu was invented by Tokujiro Namikoshi during the 1920s. However, its history goes back to the roots of massage in Japan. Shiatsu was derived from a historical Japanese massage modality known as Anma. Anma was derived from a traditional Chinese massage modality known as Tui Na. … Read More
What is Shiatsu? An Overview of Shiatsu Massage
You've likely familiar with the term Shiatsu and know that it's related to massage therapy, but don't know much more about it. In this article, we will explore Shiatsu in depth, including: The definition of Shiatsu History of Shiatsu Clinical evidence for Shiatsu Studying Shiatsu at a massage therapy school Massage Therapy career paths with Shiatsu What is Shiatsu? Shiatsu, literally meaning “figure pressure,” is a Japanese massage modality that was invented by Tokujiro Namikoshi during the 1920s. It is based on the principles of Traditional Chinese Medicine (TCM) and is considered a form of acupressure massage. … Read More
What is an Oriental Medicine Degree and What’s the ROI?
Are you considering going to acupuncture school and pursuing a degree in Oriental Medicine? Oriental Medicine is a unique health care profession that combines the holistic healing theories of Traditional Chinese Medicine (TCM) with the rigors of modern science and evidence-based medicine. Because acupuncture school tuition is expensive, you may be wondering about the benefits of becoming an acupuncturist. Well for starters, the U.S. Bureau of Labor Statistics reports that the median annual wage for Acupuncturists and Healthcare Diagnosing or Treating Practitioners, All Other from May 2020 is $82,420. This article highlights what an acupuncturist is, what they do, and the return on investment (ROI) that a Degree in Acupuncture and Oriental Medicine can provide. … Read More
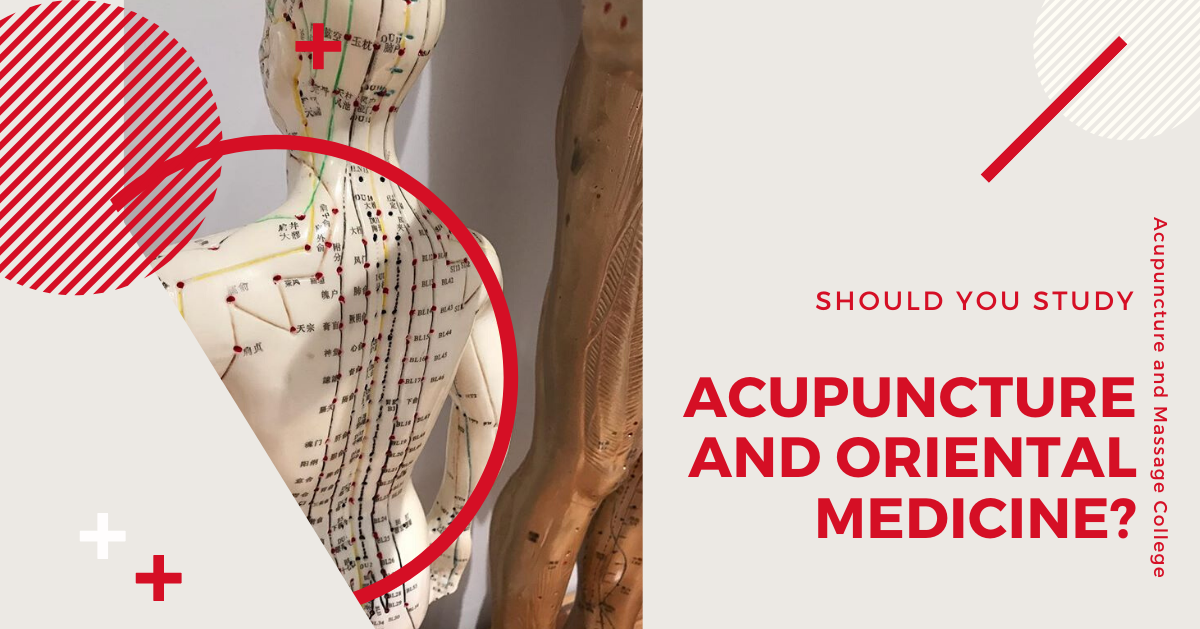
Should You Study Acupuncture and Oriental Medicine?
Are you considering going to acupuncture school? In recent years, holistic practices such as Traditional Chinese Medicine (TCM) have grown in popularity. In fact, many of the leading university hospital systems in the US now employ full-time acupuncturists. For example, Duke University’s Integrative Medicine Center offers acupuncture, acupressure massage, mindfulness meditation, and other holistic treatments in coordination with other conventional medical treatments. … Read More
How to Save on Acupuncture School Tuition
Acupuncture and Massage College in Miami, FL opened in 1983 as the first acupuncture school in Florida. For more than 30 years, we have seen countless graduates make positive changes in their personal and professional lives. … Read More
Massage Therapy Career Satisfaction Survey
The massage therapy industry is booming and many licensed massage therapists (LMTs) are enjoying the freedom to work where and when they want, and massage therapy students are enjoying job prospects like never before. However, does this mean that massage therapists are satisfied with their careers? This is what Massage Tables Now set out to uncover by surveying 1,197 professional massage therapists in the United States and Canada, to gain insights about their overall career satisfaction. … Read More
Is Acupuncture School Worth It?
If you’ve reading this article, then it’s safe to assume that you’re considering becoming an acupuncturist and enrolling in an acupuncture school in Florida. And like most prospective students, you’re concerned about the high cost of acupuncture school. … Read More
What is the Traditional Chinese Medicine (TCM) Model of the Body?
The Human Body in Traditional Chinese Medicine (TCM) In the first article of the series, we discussed the differences between Western Oriental Medicine and how Traditional Chinese Medicine (TCM) was a form of holistic medicine, since it focused on healing the physical and non-physical aspects of the human condition. In the second article, we discussed how Taoism sets the intellectual foundation for understanding TCM. Unsurprisingly, Traditional Chinese Medicine views the body as having physical and non-physical components. Knowing what these non-physical components are is important to understanding how the human body is viewed in TCM, which in turn sets the foundation for understanding TCM notions of health, illness, and treatments. As such, in this article we will give an overview of the TCM model of the human body, which is taught alongside Western anatomy at our acupuncture school in Florida. … Read More
Health and Illness in Traditional Chinese Medicine (TCM)
In the previous blog, we discussed the Traditional Chinese Medical model of the human body. In TCM, the concept of health is different than that in the West. TCM’s concept of health is based on different principles, all of which are connected to yin-yang and qi. … Read More
Eight Principles of Diagnosis in Traditional Chinese Medicine
In a previous article, we discussed the notion of health and illness in Traditional Chinese Medicine (TCM). According to TCM, illness arises as a result of specific yin-yang imbalances of the Functional Entities. … Read More
How To Research Acupuncture Schools Online
The journey to selecting the right acupuncture school for you begins by gathering information online. In this day and age, every acupuncture school should have a college website, an active social media presence (especially on Facebook), and a business profile listed in several online review directories, such as Yelp. … Read More
Should I Get a Master's or Doctorate Degree in Oriental Medicine?
A common question we get from prospective students at our acupuncture school in Florida is whether it's better to enroll in a Master's or Doctorate of Acupuncture and Oriental Medicine (AOM) Program. We understand that it’s easy and natural to become hyped when looking into different Acupuncture and Oriental Medicine degree programs. And while it is certainly good to be excited about becoming an acupuncturist, what you don’t want to do is let the paradox of choice overwhelm you, or worse, stop you completely from selecting an acupuncture school that’s right for you. … Read More

 (305) 595-9500
(305) 595-9500